Types of Energy
Energy is invisible yet it’s all around us and throughout the universe. We use it every day, we have it in our bodies and some of it comes from other planets! Energy can never be made or destroyed, but its form can be converted and changed. For example, the chemical energy we get from our food turns into kinetic and thermal energy (see below) when we walk and sound energy when we shout. Here’s a great video by scientists showing the many ways that energy can be transferred and converted.
The following chapters describe the various forms energy can take and the story behind humans, energy, fuel and the environment.
Types of energy
First let’s take a look at the different forms energy can take and how they can be converted.
 Kinetic - Anything that moves has this kind of energy. To run, cycle, climb and move the mouse for a computer we use kinetic energy.
Kinetic - Anything that moves has this kind of energy. To run, cycle, climb and move the mouse for a computer we use kinetic energy.
 Radiant - Radiant energy means light. Examples of things that have radiant energy are the Sun, lightbulbs and our computer screens. Plants convert light energy into chemical energy (food) which helps them to grown, this is known as photosynthesis.
Radiant - Radiant energy means light. Examples of things that have radiant energy are the Sun, lightbulbs and our computer screens. Plants convert light energy into chemical energy (food) which helps them to grown, this is known as photosynthesis.
 Sound - The louder we shout the more sound energy we are using. Apart from our voices there are plenty of other examples of sound energies - guitars have sound energy when plucked using kinetic energy. So do the brakes on our bikes when we’re traveling fast.
Sound - The louder we shout the more sound energy we are using. Apart from our voices there are plenty of other examples of sound energies - guitars have sound energy when plucked using kinetic energy. So do the brakes on our bikes when we’re traveling fast.
 Thermal - If you’ve ever worn thermal underwear you might guess that thermal energy is about heat! The sun gives off thermal energy, as do radiators and fires. Even lightbulbs when they’ve been on for a long time - ouch!
Thermal - If you’ve ever worn thermal underwear you might guess that thermal energy is about heat! The sun gives off thermal energy, as do radiators and fires. Even lightbulbs when they’ve been on for a long time - ouch!
 Elastic - Stretch a rubber band, or pull back the elastic on a slingshot and you’ll have elastic energy. This kind of energy is known as ‘potential energy’ as it can be stored until it’s released - usually giving way to kinetic energy!
Elastic - Stretch a rubber band, or pull back the elastic on a slingshot and you’ll have elastic energy. This kind of energy is known as ‘potential energy’ as it can be stored until it’s released - usually giving way to kinetic energy!
 Gravitational - Ever fallen out of a tree? Then you were a victim of gravitational energy. Gravitational energy is the force which keeps us on the ground. Gravitational energy is why cycling downhill is faster than cycling up hill and why ski jumpers always come back to earth.
Gravitational - Ever fallen out of a tree? Then you were a victim of gravitational energy. Gravitational energy is the force which keeps us on the ground. Gravitational energy is why cycling downhill is faster than cycling up hill and why ski jumpers always come back to earth.
 Chemical - Chemical energies come in many different forms. We get chemical energy from foods, which we use to run about, and move and talk (kinetic and sound energy). Chemical energies are stored in fuels which we burn to release thermal energy - this is one way of making electricity, see Electricity for more information.
Chemical - Chemical energies come in many different forms. We get chemical energy from foods, which we use to run about, and move and talk (kinetic and sound energy). Chemical energies are stored in fuels which we burn to release thermal energy - this is one way of making electricity, see Electricity for more information.
 Nuclear - Nuclear energy is the energy stored inside tiny atoms. Atoms are invisible and make up the elements of the whole universe! There are three parts to these tiny particles which are - protons, electrons and neutrons. Nuclear energy is released when atoms are joined together (fusion) or split apart (fission). Doing this converts energy into thermal and radiant energy. The Sun uses nuclear energy to produce its light and heat.
Nuclear - Nuclear energy is the energy stored inside tiny atoms. Atoms are invisible and make up the elements of the whole universe! There are three parts to these tiny particles which are - protons, electrons and neutrons. Nuclear energy is released when atoms are joined together (fusion) or split apart (fission). Doing this converts energy into thermal and radiant energy. The Sun uses nuclear energy to produce its light and heat.
 Converting Energy
Converting Energy
When we go for a run we are using chemical energy in our bodies to produce movement (kinetic energy), which in turn converts to warmth (thermal energy). Cycling downhill involves kinetic and gravitational energy. The friction between the tyres and the road will also cause the tyres to warm up a little producing thermal energy. What form will some of this energy take when putting on the brakes?
Fuel For Humans
All living things need energy from food and/or water to survive. But, unlike most living things we humans want energy in lots of other ways too.
 When humans first roamed the earth we kept warm using animal skins, and sheltered in caves or houses built with plants or mud. Later, and not so long ago we lit our houses with candles, cooked our food on open fires and travelled by horse and cart or by foot. Nowadays we use electric light bulbs, electric or gas ovens for cooking and petrol/diesel powered cars to get around. These new devices and inventions require a lot of energy and so need plenty of fuel to make that energy.
When humans first roamed the earth we kept warm using animal skins, and sheltered in caves or houses built with plants or mud. Later, and not so long ago we lit our houses with candles, cooked our food on open fires and travelled by horse and cart or by foot. Nowadays we use electric light bulbs, electric or gas ovens for cooking and petrol/diesel powered cars to get around. These new devices and inventions require a lot of energy and so need plenty of fuel to make that energy.
Most of the energy that we need is in the form of electricity, gas or petrol/diesel. Let’s take a look at these energy supplies to learn more about what they are and how they work...
Electricity 1
 Electrical energy is natural and can be seen as strikes of lightning during thunder storms. Humans have been fascinated with electricity for hundreds of years, and have slowly gained more and more understanding of this bright and powerful energy.
Electrical energy is natural and can be seen as strikes of lightning during thunder storms. Humans have been fascinated with electricity for hundreds of years, and have slowly gained more and more understanding of this bright and powerful energy.
History
The first power station in UK began generating electricity for mass consumption in London, in 1882. This invention accelerated the Industrial Revolution and the powered-up way we live today.
How is this electricity generated?
Small amounts of electrical energy can be generated by spinning a magnet within a coil of wire. But to generate enough electricity for a whole city giant machines called ‘generators’ at power stations need to be used. Over the years we have found many different ways to ‘fuel’ these generators.
In many power stations, non-renewable fossil fuels such as oil, coal and gas are burnt to create heat. Others burn recently living organisms - known as biomass. Some use heat that occurs on its own underground, this is called geothermal energy. Let’s look at the processes which turn these fuels into electricity!
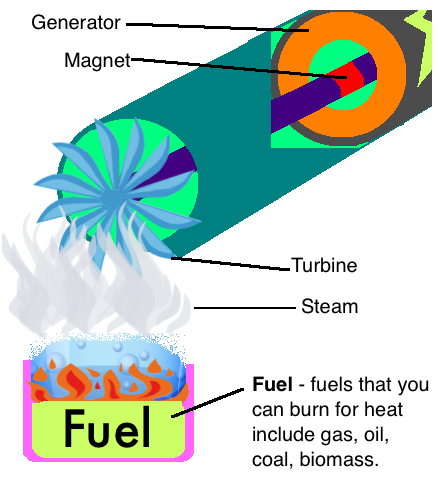 First the fuel is burnt to provide heat
First the fuel is burnt to provide heat- This heat is used to boil water
- Steam from the boiling water rises upwards into turbines
- The pressure of the steam provides enough kinetic energy to turn the blades of the turbines
- The movement of the turbines turns the magnet in the generator producing electricity
Wind, tidal and hydro-electric power supplies also have generators and spinning blades but they do not require heat to turn the turbines. Can you guess how each of them turn their turbines?
Here is a list of fuels used for their chemical energies to produce electricity, some are renewable, some aren’t-
Oil (also known as crude oil or petroleum)
Coal
Gas
Peat
Uranium (nuclear)
Biomass
Biofuels
Energies used in producing electricity -
Geothermal (thermal energy)
Solar power (radiant energy)
Wind (kinetic energy)
Tidal (Kinetic and gravitational energy)
Hydroelectric (kinetic energy)
Fossil fuels or non-renewable energies?
 Fossil fuels are so-called because they formed underground over millions and millions of years. Because these fuels take so long to form, once we use up all the fossil fuels we won’t be able to replace them! That is why they are known as non-renewable. The amount of these fuels that we use has increased dramatically across the world and sooner or later they will run out. This is why renewable energy is a big part of the planet’s future.
Fossil fuels are so-called because they formed underground over millions and millions of years. Because these fuels take so long to form, once we use up all the fossil fuels we won’t be able to replace them! That is why they are known as non-renewable. The amount of these fuels that we use has increased dramatically across the world and sooner or later they will run out. This is why renewable energy is a big part of the planet’s future.
Electricity 2
 Renewable energies harness very different kinds of fuel to create electricity. Examples of this include using radiant energy from the sun to fuel solar panels. Wind turbines use the wind and tidal power use waves and the power of the tides in the sea.
Renewable energies harness very different kinds of fuel to create electricity. Examples of this include using radiant energy from the sun to fuel solar panels. Wind turbines use the wind and tidal power use waves and the power of the tides in the sea.
So there are many ways of making electricity, and many fuels to make this happen. We depend on electricity to power our homes, schools, workplaces and shops - indeed it powers whole countries!
How much electricity does the world use?
So, how much electricity does the whole world use? First we have to learn a new word - TWh means terawatt hours. This measurement unit is used when measuring mass energy production and consumption. Just one terawatt is equal to one trillion watts (1,000,000,000,000). (An energy saving lightbulb requires just 20 watts or less per hour) Electricity continues to position itself as the “fuel” of the future, with global electricity demand growing by 4% in 2018. So, to answer the question, the amount of electricity generated by the world each year has risen to more than 23 000 TWh! In 1971 the world total was just 5,000 TWh. (see source below)
Remember that electricity is like a river because when it is in use it is flowing through electrical cables very fast and being converted into other types of energy such as radiant energy in your lightbulb. The electricity that powered your computer ten minutes, or ten seconds ago isn’t the same electricity powering it now because, like water in a river, electricity is alway flowing - this is why your parents get electricity bills!
Environment
Unfortunately, to make so much energy we also make a lot of pollution in the process. Some fuels are better for the environment than others.
Coal and Oil
The burning of fossil fuels not only generates electrical energy but produces a lot of chemical energy too in the form of emissions and pollution. The fossil fuels oil and coal are mostly made up of carbon. When we burn them this carbon is released into the atmosphere where it mixes with oxygen in the air to form carbon dioxide - also known as CO2. CO2 acts like a blanket covering the earth, trapping the sun’s warmth and heating up the planet (this is known as the greenhouse effect). Our growing demand for energy, coupled with an ever increasing human population means that the level of CO2 in our atmosphere is always increasing.
To reduce the impact of climate change people all over the world are thinking of new and better ways to make electricity, power cars and create a sustainable future. Everyone can do their bit to help use less electricity and petrol. To find out more see the Saving Energy section.
 Don’t forget that the finding and collecting of fuels such as oil and coal also creates many negative impacts on local and global environments. For example drilling for oil in rainforests means that thousands of trees are cut down, animals and humans lose their habitats and ecosystems are polluted with oil.
Don’t forget that the finding and collecting of fuels such as oil and coal also creates many negative impacts on local and global environments. For example drilling for oil in rainforests means that thousands of trees are cut down, animals and humans lose their habitats and ecosystems are polluted with oil.
Solutions
Solutions to the problem of fossils fuel emissions and the damage being done to habitats and the environment mostly lie in the future of renewable technology. Another non-renewable solution which is being explored by many countries at the moment is the controversial method of hydraulic fracturing - better known as fracking.
It is clear that without energy our towns and cities and indeed our phones, mp3 players, kettles and toasters would stop working! The only way to make this energy cleaner is to develop renewable technologies that don’t harm the earth and to reduce our demand for this energy.
If you would like to know what you can do to help (even switching off your lights while you’re not in your room is helping the planet!) then take a look at our ‘Environment’ factsheet below and become an energy saver today!
1. International Energy Agency - https://www.iea.org/reports/key-world-energy-statistics-2019
Gas
Gas is short for ‘natural gas’, an invisible, odourless substance. It is found underground and is a fossil fuel. Gas is used as a fuel in our homes, in industry and in the energy sectors to produce electricity, heat buildings, water and cook food.
Natural gas is made up of a mixture of gases but methane is the main ingredient. Although natural gas itself does not smell, gas companies add a chemical to it that produces an eggy rotten smell so that we can easily sniff out any dangerous gas leaks.
History
It is said that the Chinese used gas to heat water, transporting it from open wells through the hollow stems of bamboo as long ago as 500 BC. Yet, it was more than 1500 years before natural gas became a mainstream energy supply. In 1795 the UK became the first country to commercialise (produce and sell) natural gas. Confusingly this natural gas was not ‘naturally occuring’, but gas produced from coal. Initially gas was used only for lighting street lamps, but these were later replaced by more efficient electric lamps. In 1885 the invention of the infamous Bunsen burner changed the use of gas throughout the world and preceded the development of the infrastructure (pipes, stations and machines) needed to extract, transport and provide gas to homes and businesses across the country.

Gas in the home
Homes and workplaces in the UK use gas for many different things. Some houses have gas hobs and ovens for cooking. Some heat their house and water using gas boilers. The benefits of gas are that it burns hot, bright and does not smoke when burnt like other fuels.
Environment
Like coal and oil, burning gas to produce electricity produces carbon dioxide and methane emissions - two ‘greenhouse gases’. However, gas produces considerably less emissions than oil and coal making it a cleaner alternative. Despite this advantage gas is a non-renewable fossil fuel which means that there is only a limited supply.
Fracking
In a bid to find and extract more gas from beneath the earth’s surface methods have become increasingly complicated. The most recent, and controversial, method is hydraulic fracturing, also known as ‘fracking’. Fracking is the extraction of shale gas from layers of rock and sediment deep underground. To extract the gas various chemicals and a lot of water are pumped into the ground, squeezing out the gas as it fills up the holes. Many people are concerned that fracking causes earthquakes and that the chemicals used in the process can leak into our water supplies.
Tara, aged 11 recorded a video for us about the hydraulic fracturing that was going on around her home, you can view it here.
Petrol and Diesel
Petrol and diesel come from crude oil, which comes from deep underground. Crude oil is refined to make petrol (in America this is known as gasoline) or diesel.
Most forms of transport use either petrol or diesel to power their engines - from lawnmowers, cars, buses and motorbikes to large ships and aeroplanes. Petrol and diesel power the lorries and ships that get food to the supermarket, cars and buses might help you get to school, in fact, petrol and diesel make so many things possible that, like electricity, they’re now vital to our way of life!
History
The history of crude oil starts between 150 and 200 million years ago when animals living in the sea - plankton and other organisms - died and sank to the sea bed. Over time, more and more bodies of sea creature built up on the sea bed and were covered over with silt. The silt built up more and more and started to turn into rock. Meanwhile, the bodies of the plankton and other animals, trapped under the silt, were changed by pressure and heat of between 60 and 120 degrees C into the black, goopy liquid we call crude oil. Large deposits of oil can be found deep underground and under the sea bed in different parts of the world.
 The history of petrol and diesel as a transportation fuel for humans began in 1858. The first combustion engine was invented by an engineer from Belgium but it took another 40 years before petrol-powered cars were sold commercially. American industrialist Henry Ford is famous for inventing the assembly line technique of mass production that paved the way for millions of factories and products. Henry Ford of the Ford Motoring Company began to sell the Model T ford in 1908 and went on to sell 15 million in the next twenty years. This also led to the creation of miles and miles of roads and streets across the USA. Ford are still operating today, although the cars look a little different.
The history of petrol and diesel as a transportation fuel for humans began in 1858. The first combustion engine was invented by an engineer from Belgium but it took another 40 years before petrol-powered cars were sold commercially. American industrialist Henry Ford is famous for inventing the assembly line technique of mass production that paved the way for millions of factories and products. Henry Ford of the Ford Motoring Company began to sell the Model T ford in 1908 and went on to sell 15 million in the next twenty years. This also led to the creation of miles and miles of roads and streets across the USA. Ford are still operating today, although the cars look a little different.
How it works
An engine’s purpose is to convert the chemical energy of the fuel into kinetic energy, movement! Petrol or diesel is burnt in the internal combustion engines of vehicles. Like power stations, engines use heat and pressure to move moving parts that drive the car forwards.
Byproducts
Unfortunately, as well as kinetic, sound and thermal energy this combustion technique also produces chemical energy as a byproduct. This chemical energy leaves the engine via the exhaust pipe as exhaust fumes. Sadly, many of these fumes are harmful for us, animals and the environment. Let’s take a look at the different chemicals you might find in the fumes -
- Carbon dioxide (CO2) is a greenhouse gas and contributes to global warming
- Carbon monoxide (CO) is a poisonous gas
- Sulphur dioxide (SO2) is a cause of acid rain and can cause breathing or heart problems
- Nitrogen oxides (NOX) and Volatile Organic Compounds (VOC) can irritate and damage the lungs
- Particulate matter (PM) is what makes cities hazy and foggy. It contributes to ground-level ozone (a gas which is good for the planet when it is up above the clouds, but bad for us when it is at ground-level) These particles also contribute to asthma and other lung problems.
- Lead is poisonous but only in extremely high doses. Various airborne toxins such as benzene, formaldehyde, acetaldehyde can also cause health problems. These chemicals also contribute to ground-level ozone.
Solutions
Alternatives to petrol and diesel are well underway and include electric cars and biodiesel. Some cars can be converted to run on the oil used to cook chips! Others can be powered by plants that are refined in factories into liquids a little bit like petrol. But these solutions also create problems of their own. Electric cars obviously need electricity and how ‘clean’ or green the cars are also depends on how that electricity is produced.
Biodiesels on the other hand require acres and acres of crops which are often grown where lush primary rainforests once stood. The amount of land needed to grow crops for the fuel could be left as rainforest or, if it was already clear, could be helpful for growing food for our ever-growing population, or the world’s livestock.
For more information on oil and petroleum products see Energy Kids.
Batteries
 Contrary to popular belief, a battery does not store energy but it is used to make electrical energy when needed.
Contrary to popular belief, a battery does not store energy but it is used to make electrical energy when needed.
Electrical energy is produced in a battery by the reactions of two electrodes. Each electrode is linked to one of the battery’s metal terminals. When the battery is linked into a circuit, there is a flow of electrons from one terminal (negative) through the circuit to the other terminal (positive). This is because the electrode starts to dissolve releasing electrons into the wire of the circuit at the negative terminal. The other electrode also loses electrons from one electrode through the circuit to make up the deficiency at the other electrode. In this way chemical energy is converted into electrical energy. Some batteries can be recharged by passing electrical energy through them. The electrical energy reverses the chemical reactions that produce the power.
Activity: A simple battery can be made by using a pile of ten 2p coins interspaced with blotting paper and foil. Cut our some round shapes of blotting paper and aluminium foil (a little larger than a 2p coin). Soak the blotting paper in salty water. Place a piece of foil over a 2p coin and a piece of salty blotting paper on top. Repeat these layers to make a small pile. Fix the end of a piece of wire underneath the pile and another on top. If the free ends of the wire are connected in a circuit with a light bulb, chemical reactions in the pile make electrical energy flow and the bulb lights up.
Saving Energy
When you’re next using electricity think about where it comes from and how you can use less to take the strain off of the environment - here are some simple ways to make a big difference:
- On or Off? Leaving televisions, stereos and computers on standby with the little red light showing still uses up electricity, even though they might look like they're turned off. To stop this you can turn them off at the wall. Simple!
- Lights out! Turning lights off when you're not in the room (make sure no-one else is too!) can save a lot of energy.
- Putting the kettle on In the UK we drink approximately 100 million cups of tea daily, which is almost 36 billion per year according to the Tea Council. That's a lot of tea, and a lot of electricity too! That's why it's important to only boil as much water as you need in your kettle. It's best to boil enough water for one cup, not three or five - plus you won't have to wait as long, which is a bonus!
- Brrrr The fridge is an important part of the kitchen, it keeps food fresh, cool and healthy! But it takes a lot of energy to keep cool, so help it by not leaving the fridge door open or putting hot things inside.
- Shopping Many products are now helping you to reduce your energy use and your parents’ energy bills! Check new products for their efficiency grade and rating and keep an eye out for the Energy Efficiency Recommended logo.
- Harness the sun’s energy - An increasing number of people are fitting solar panels to their houses. Photovoltaic (PV) solar panels create electricity from sunlight, which can then be used to power their homes. Solar heating panels make hot water, reducing the need for electricity or gas to heat water or run the central heating system.

Helpful websites - Energy kids and Energy World.
Energy Activities
Imagine a week without electricity and/or fuel, write a diary - how would life be different?
Guess how many things in your house use electricity, from microwaves to mobile phone chargers, then go to each room and count them up. Was your guess close to the real figure?
Think about how we used energy 200 years ago - what energies did we use? What energies do we use now? How do these compare and how do they effect the planet and its natural resources?
Credits
Image: Energy by Brian R Jeffery
 Kinetic - Anything that moves has this kind of energy. To run, cycle, climb and move the mouse for a computer we use kinetic energy.
Kinetic - Anything that moves has this kind of energy. To run, cycle, climb and move the mouse for a computer we use kinetic energy. Radiant - Radiant energy means light. Examples of things that have radiant energy are the Sun, lightbulbs and our computer screens. Plants convert light energy into chemical energy (food) which helps them to grown, this is known as photosynthesis.
Radiant - Radiant energy means light. Examples of things that have radiant energy are the Sun, lightbulbs and our computer screens. Plants convert light energy into chemical energy (food) which helps them to grown, this is known as photosynthesis.  Sound - The louder we shout the more sound energy we are using. Apart from our voices there are plenty of other examples of sound energies - guitars have sound energy when plucked using kinetic energy. So do the brakes on our bikes when we’re traveling fast.
Sound - The louder we shout the more sound energy we are using. Apart from our voices there are plenty of other examples of sound energies - guitars have sound energy when plucked using kinetic energy. So do the brakes on our bikes when we’re traveling fast. Thermal - If you’ve ever worn thermal underwear you might guess that thermal energy is about heat! The sun gives off thermal energy, as do radiators and fires. Even lightbulbs when they’ve been on for a long time - ouch!
Thermal - If you’ve ever worn thermal underwear you might guess that thermal energy is about heat! The sun gives off thermal energy, as do radiators and fires. Even lightbulbs when they’ve been on for a long time - ouch! Elastic - Stretch a rubber band, or pull back the elastic on a slingshot and you’ll have elastic energy. This kind of energy is known as ‘potential energy’ as it can be stored until it’s released - usually giving way to kinetic energy!
Elastic - Stretch a rubber band, or pull back the elastic on a slingshot and you’ll have elastic energy. This kind of energy is known as ‘potential energy’ as it can be stored until it’s released - usually giving way to kinetic energy! Gravitational - Ever fallen out of a tree? Then you were a victim of gravitational energy. Gravitational energy is the force which keeps us on the ground. Gravitational energy is why cycling downhill is faster than cycling up hill and why ski jumpers always come back to earth.
Gravitational - Ever fallen out of a tree? Then you were a victim of gravitational energy. Gravitational energy is the force which keeps us on the ground. Gravitational energy is why cycling downhill is faster than cycling up hill and why ski jumpers always come back to earth. Chemical - Chemical energies come in many different forms. We get chemical energy from foods, which we use to run about, and move and talk (kinetic and sound energy). Chemical energies are stored in fuels which we burn to release thermal energy - this is one way of making electricity, see Electricity for more information.
Chemical - Chemical energies come in many different forms. We get chemical energy from foods, which we use to run about, and move and talk (kinetic and sound energy). Chemical energies are stored in fuels which we burn to release thermal energy - this is one way of making electricity, see Electricity for more information. Nuclear - Nuclear energy is the energy stored inside tiny atoms. Atoms are invisible and make up the elements of the whole universe! There are three parts to these tiny particles which are - protons, electrons and neutrons. Nuclear energy is released when atoms are joined together (fusion) or split apart (fission). Doing this converts energy into thermal and radiant energy. The Sun uses nuclear energy to produce its light and heat.
Nuclear - Nuclear energy is the energy stored inside tiny atoms. Atoms are invisible and make up the elements of the whole universe! There are three parts to these tiny particles which are - protons, electrons and neutrons. Nuclear energy is released when atoms are joined together (fusion) or split apart (fission). Doing this converts energy into thermal and radiant energy. The Sun uses nuclear energy to produce its light and heat. Converting Energy
Converting Energy
 When humans first roamed the earth we kept warm using animal skins, and sheltered in caves or houses built with plants or mud. Later, and not so long ago we lit our houses with candles, cooked our food on open fires and travelled by horse and cart or by foot. Nowadays we use electric light bulbs, electric or gas ovens for cooking and petrol/diesel powered cars to get around. These new devices and inventions require a lot of energy and so need plenty of fuel to make that energy.
When humans first roamed the earth we kept warm using animal skins, and sheltered in caves or houses built with plants or mud. Later, and not so long ago we lit our houses with candles, cooked our food on open fires and travelled by horse and cart or by foot. Nowadays we use electric light bulbs, electric or gas ovens for cooking and petrol/diesel powered cars to get around. These new devices and inventions require a lot of energy and so need plenty of fuel to make that energy. Electrical energy is natural and can be seen as strikes of lightning during thunder storms. Humans have been fascinated with electricity for hundreds of years, and have slowly gained more and more understanding of this bright and powerful energy.
Electrical energy is natural and can be seen as strikes of lightning during thunder storms. Humans have been fascinated with electricity for hundreds of years, and have slowly gained more and more understanding of this bright and powerful energy.  First the fuel is burnt to provide heat
First the fuel is burnt to provide heat Fossil fuels are so-called because they formed underground over millions and millions of years. Because these fuels take so long to form, once we use up all the fossil fuels we won’t be able to replace them! That is why they are known as non-renewable. The amount of these fuels that we use has increased dramatically across the world and sooner or later they will run out. This is why renewable energy is a big part of the planet’s future.
Fossil fuels are so-called because they formed underground over millions and millions of years. Because these fuels take so long to form, once we use up all the fossil fuels we won’t be able to replace them! That is why they are known as non-renewable. The amount of these fuels that we use has increased dramatically across the world and sooner or later they will run out. This is why renewable energy is a big part of the planet’s future. Renewable energies harness very different kinds of fuel to create electricity. Examples of this include using radiant energy from the sun to fuel solar panels. Wind turbines use the wind and tidal power use waves and the power of the tides in the sea.
Renewable energies harness very different kinds of fuel to create electricity. Examples of this include using radiant energy from the sun to fuel solar panels. Wind turbines use the wind and tidal power use waves and the power of the tides in the sea.  Don’t forget that the finding and collecting of fuels such as oil and coal also creates many negative impacts on local and global environments. For example drilling for oil in rainforests means that thousands of trees are cut down, animals and humans lose their habitats and ecosystems are polluted with oil.
Don’t forget that the finding and collecting of fuels such as oil and coal also creates many negative impacts on local and global environments. For example drilling for oil in rainforests means that thousands of trees are cut down, animals and humans lose their habitats and ecosystems are polluted with oil.
 The history of petrol and diesel as a transportation fuel for humans began in 1858. The first combustion engine was invented by an engineer from Belgium but it took another 40 years before petrol-powered cars were sold commercially. American industrialist Henry Ford is famous for inventing the assembly line technique of mass production that paved the way for millions of factories and products. Henry Ford of the Ford Motoring Company began to sell the Model T ford in 1908 and went on to sell 15 million in the next twenty years. This also led to the creation of miles and miles of roads and streets across the USA. Ford are still operating today, although the cars look a little different.
The history of petrol and diesel as a transportation fuel for humans began in 1858. The first combustion engine was invented by an engineer from Belgium but it took another 40 years before petrol-powered cars were sold commercially. American industrialist Henry Ford is famous for inventing the assembly line technique of mass production that paved the way for millions of factories and products. Henry Ford of the Ford Motoring Company began to sell the Model T ford in 1908 and went on to sell 15 million in the next twenty years. This also led to the creation of miles and miles of roads and streets across the USA. Ford are still operating today, although the cars look a little different. Contrary to popular belief, a battery does not store energy but it is used to make electrical energy when needed.
Contrary to popular belief, a battery does not store energy but it is used to make electrical energy when needed.
